Retatrutide for Weight Loss: The Triple-Action Treatment Redefining What’s Possible
Physician-Supervised Retatrutide Therapy — Personalized Plans, Measurable Results
Retatrutide is a next-generation GIP, GLP-1, and glucagon triple-receptor agonist that clinical trials have shown can help patients lose significantly more weight than any single-target GLP-1 medication. At Regal Weight Loss, our licensed medical providers create individualized retatrutide treatment plans designed around your health history, goals, and lifestyle — so you get the most from one of the most powerful weight loss therapies available today.
What Is Retatrutide?
Retatrutide is an investigational injectable weight loss medication developed by Eli Lilly that works by simultaneously activating three hormone receptors in the body: GIP (glucose-dependent insulinotropic polypeptide), GLP-1 (glucagon-like peptide-1), and glucagon. This triple-receptor mechanism is what separates retatrutide from every other weight loss medication currently available — including popular GLP-1 medications like semaglutide (Ozempic, Wegovy) and the dual-receptor medication tirzepatide (Mounjaro, Zepbound).
Because retatrutide targets all three receptors simultaneously, it addresses weight loss through multiple biological pathways at once — reducing appetite, improving insulin sensitivity, increasing metabolic rate, and promoting the breakdown of stored fat. In Phase 2 clinical trials published in The New England Journal of Medicine, participants taking the highest dose of retatrutide lost an average of 24.2% of their body weight over 48 weeks — a result that has not been matched by any other weight loss medication studied to date.
Retatrutide is currently in Phase 3 clinical trials and is not yet FDA-approved as a standalone medication. However, it may be available through licensed medical providers via compounding pharmacies while Phase 3 trials are ongoing. At Regal Weight Loss, our physicians stay at the forefront of emerging obesity medicine to ensure our patients have access to the most advanced, evidence-based treatment options available.
How Does Retatrutide Work?
Retatrutide works through a mechanism called triple-receptor agonism — meaning it activates three separate hormone receptors that each play a distinct role in how your body manages hunger, blood sugar, and fat storage. Most weight loss medications target only one of these pathways. Retatrutide targets all three simultaneously, which is why its clinical results have been so significantly greater than anything that has come before it.
GLP-1 Receptor Activation — Appetite and Blood Sugar Control
The GLP-1 (glucagon-like peptide-1) receptor is the same target used by semaglutide and tirzepatide. When activated, it signals the brain to reduce hunger and cravings, slows the rate at which the stomach empties food into the intestines, and helps the pancreas regulate insulin production in response to meals. This is the primary mechanism responsible for the reduced appetite most patients experience on GLP-1 based therapies.
GIP Receptor Activation — Enhanced Metabolic Response
The GIP (glucose-dependent insulinotropic polypeptide) receptor is also targeted by tirzepatide, but not by semaglutide. GIP activation enhances the body’s insulin response after eating, improves how efficiently cells absorb and use glucose, and has been shown to amplify the appetite-suppressing effects of GLP-1 activation. The combination of GLP-1 and GIP receptor agonism is what made tirzepatide such a significant advancement over semaglutide — and retatrutide builds on that foundation further.
Glucagon Receptor Activation — The Differentiator
The glucagon receptor is what makes retatrutide truly unique. Glucagon is a hormone that signals the liver to release stored energy and increases the body’s overall metabolic rate — essentially instructing the body to burn more calories at rest. While glucagon activation in isolation can raise blood sugar, retatrutide is engineered so that the simultaneous GLP-1 and GIP activation counterbalances this effect, keeping blood sugar stable while still capturing the fat-burning and metabolism-boosting benefits of glucagon signaling. No other approved or late-stage weight loss medication activates the glucagon receptor in this way.
The result of activating all three receptors together is a compounding effect that addresses the biological roots of obesity more comprehensively than any single or dual-receptor approach. Patients experience reduced hunger, improved blood sugar regulation, a higher resting metabolic rate, and accelerated breakdown of stored fat — all simultaneously. At Regal Weight Loss, our physicians use this understanding of retatrutide’s mechanism to personalize dosing protocols that maximize each patient’s results while carefully monitoring for side effects.
Retatrutide vs. Semaglutide vs. Tirzepatide: How Do They Compare?
The landscape of weight loss medication has evolved dramatically over the past several years. Semaglutide set a new standard, tirzepatide raised it further, and now retatrutide is emerging from clinical trials with results that suggest it may represent the most significant leap forward yet. Understanding how these three medications differ — in mechanism, clinical outcomes, and patient experience — is essential for making an informed decision about which therapy is right for you.
Comparison Table:
| Semaglutide | Tirzepatide | Retatrutide | |
|---|---|---|---|
| Brand Names | Ozempic, Wegovy | Mounjaro, Zepbound | Investigational (no brand name yet) |
| Receptor Targets | GLP-1 only | GLP-1 + GIP | GLP-1 + GIP + Glucagon |
| Administration | Weekly injection | Weekly injection | Weekly injection |
| Avg. Weight Loss (clinical trials) | ~15% body weight | ~20% body weight | ~24% body weight |
| FDA Approval Status | Approved | Approved | Phase 3 trials ongoing |
| Available at Regal Weight Loss | Yes | Yes | Yes (via licensed compounding) |
Retatrutide vs. Semaglutide
Semaglutide was the first GLP-1 medication to demonstrate double-digit percentage weight loss in clinical trials, and it remains one of the most widely prescribed weight loss medications in the world. However, semaglutide targets only the GLP-1 receptor, meaning it addresses hunger and blood sugar regulation but does not engage the GIP or glucagon pathways. In the STEP trials, semaglutide produced an average weight loss of approximately 15% of body weight over 68 weeks — a landmark result at the time. Retatrutide’s Phase 2 trials produced an average of 24.2% body weight loss over 48 weeks — a shorter timeframe with a significantly greater outcome. For patients who have plateaued on semaglutide or are looking for a more aggressive approach to weight loss, retatrutide represents a meaningful step forward in both mechanism and results.
Retatrutide vs. Tirzepatide
Tirzepatide was a major advancement over semaglutide because it added GIP receptor activation to GLP-1, creating a dual-receptor effect that produced superior weight loss outcomes in the SURMOUNT trials — approximately 20% body weight reduction. Retatrutide builds directly on tirzepatide’s dual-receptor foundation by adding glucagon receptor activation as a third pathway, which increases resting metabolic rate and accelerates fat breakdown in ways that tirzepatide cannot. The clinical trial data suggests this third receptor is responsible for the additional 4–5% average weight loss advantage retatrutide holds over tirzepatide. For patients who have done well on tirzepatide but want to explore whether a triple-receptor approach could deliver greater results, retatrutide is the most logical next step.
Which Medication Is Right for You?
The best weight loss medication is the one that is right for your individual health profile, medical history, and goals — and that determination should always be made in partnership with a licensed medical provider. At Regal Weight Loss, our physicians evaluate each patient comprehensively before recommending any treatment, including retatrutide. Factors such as your current weight, metabolic health, prior medication history, and tolerance for injectable therapies all play a role in determining which approach will deliver the safest and most effective results for you specifically.
Not sure which medication is right for you? Schedule a consultation with a Regal Weight Loss physician and get a personalized recommendation based on your health history and goals.
Retatrutide Clinical Trial Results: What the Data Shows
The clinical evidence behind retatrutide is what sets it apart from every other weight loss medication currently available. In a landmark Phase 2 trial published in The New England Journal of Medicine in 2023, retatrutide produced weight loss results that exceeded anything previously recorded in a randomized controlled trial for an anti-obesity medication. Below is a detailed breakdown of what the data showed — and what it means for patients considering retatrutide therapy.
The Phase 2 Trial: Study Design
The Phase 2 TRIUMPH trial enrolled 338 adults with obesity (BMI ≥ 30) or overweight (BMI ≥ 27) with at least one weight-related comorbidity such as hypertension, dyslipidemia, or type 2 diabetes. Participants were randomly assigned to receive weekly subcutaneous injections of retatrutide at doses of 1mg, 4mg, 8mg, or 12mg, or a placebo, over a 48-week period. The trial was double-blind and placebo-controlled — the gold standard for evaluating medication efficacy and safety.
Key Efficacy Outcomes
The results across all active dose groups were statistically significant and clinically meaningful:
- 1mg dose: Average body weight reduction of approximately 8.7% over 48 weeks
- 4mg dose: Average body weight reduction of approximately 17.5% over 48 weeks
- 8mg dose: Average body weight reduction of approximately 22.8% over 48 weeks
- 12mg dose: Average body weight reduction of approximately 24.2% over 48 weeks
- Placebo group: Average body weight reduction of approximately 2.1% over 48 weeks
Critically, the weight loss curve in the highest dose groups had not plateaued by week 48 — meaning the full extent of retatrutide’s efficacy may not yet be fully captured in Phase 2 data alone. Phase 3 trials are expected to provide longer-duration outcomes data.
Secondary Outcomes and Metabolic Benefits
Beyond raw weight loss, the Phase 2 trial documented a range of meaningful metabolic improvements across active dose groups:
- Waist circumference reduced by an average of up to 18.9 cm in the highest dose group
- Fasting insulin levels improved significantly across all active dose groups
- Blood pressure reductions were observed, particularly systolic blood pressure
- Triglyceride levels decreased substantially, a key marker of cardiovascular risk reduction
- HbA1c (a measure of long-term blood sugar control) improved in participants with elevated baseline levels
These secondary outcomes are clinically significant because they demonstrate that retatrutide’s benefits extend well beyond weight loss — addressing several of the metabolic conditions most commonly associated with obesity.
How Do These Results Compare to Other Medications?
To understand the magnitude of retatrutide’s Phase 2 results, context is essential:
To understand the magnitude of retatrutide’s Phase 2 results, context is essential:
| Medication | Trial | Duration | Avg. Weight Loss |
|---|---|---|---|
| Semaglutide 2.4mg | STEP 1 | 68 weeks | ~15% |
| Tirzepatide 15mg | SURMOUNT-1 | 72 weeks | ~20% |
| Retatrutide 12mg | Phase 2 TRIUMPH | 48 weeks | ~24.2% |
Retatrutide achieved its results in a shorter timeframe than both semaglutide and tirzepatide trials, which makes the comparison even more striking. While cross-trial comparisons must be interpreted carefully due to differences in study populations and design, the magnitude of difference is difficult to dismiss.
Where Is Retatrutide Now? Phase 3 Trial Status
Following the compelling Phase 2 results, Eli Lilly initiated the Phase 3 TRIUMPH trial program for retatrutide. Phase 3 trials involve larger patient populations, longer durations, and broader safety monitoring — and are the final regulatory step before FDA approval consideration. While a specific FDA approval timeline has not been confirmed, the Phase 3 program is actively enrolling and retatrutide remains one of the most closely watched drugs in the obesity medicine pipeline. At Regal Weight Loss, we actively monitor trial developments and regulatory updates so our patients always have access to the most current information.
The clinical data on retatrutide represents the most promising evidence base in the history of pharmaceutical weight loss treatment. For patients who have struggled to achieve meaningful results with diet, exercise, or prior medications, these numbers offer a genuinely new level of possibility. Our physicians at Regal Weight Loss are available to review this evidence with you personally and help you determine whether retatrutide is an appropriate option for your individual health profile.
Ready to discuss whether retatrutide is right for you?
Who Is a Good Candidate for Retatrutide?
Retatrutide is not a one-size-fits-all solution, and like all medical treatments, it is most effective when matched to the right patient. At Regal Weight Loss, our physicians conduct a thorough evaluation before recommending retatrutide to ensure it is both safe and appropriate for each individual. The following criteria reflect the patient profile most likely to benefit from retatrutide therapy based on clinical trial enrollment standards and current medical guidance on obesity pharmacotherapy.
You May Be a Good Candidate for Retatrutide If:
- Your BMI is 30 or higher — or 27 or higher with at least one weight-related health condition such as high blood pressure, high cholesterol, sleep apnea, or type 2 diabetes
- You have struggled to achieve or maintain significant weight loss through diet and exercise alone
- You have tried other GLP-1 medications such as semaglutide or tirzepatide and either did not achieve your goal weight or experienced diminishing results over time
- You do not have a personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2), as these are contraindications shared across GLP-1 class medications
- You are not currently pregnant or planning to become pregnant, as the safety of retatrutide during pregnancy has not been established
- You are prepared to commit to a comprehensive treatment plan that includes medical supervision, regular check-ins, and lifestyle support alongside medication
Retatrutide May Be Especially Well-Suited for You If:
- You have plateaued on tirzepatide and are looking for a next-level therapeutic option
- You have significant metabolic comorbidities such as insulin resistance, elevated triglycerides, or non-alcoholic fatty liver disease (NAFLD), as retatrutide’s glucagon activation offers particular metabolic benefits in these cases
- You have a higher starting BMI (40+) and need a medication with the clinical evidence to support substantial, sustained weight loss
- You are motivated by the most advanced evidence-based option currently available and want access to a treatment that is at the forefront of obesity medicine
Retatrutide May Not Be Right for You If:
- You have a personal or family history of medullary thyroid carcinoma or MEN 2 syndrome
- You have a history of pancreatitis or conditions associated with elevated pancreatic enzyme levels
- You are currently pregnant, breastfeeding, or planning pregnancy in the near term
- You have severe gastrointestinal conditions that may be exacerbated by medications that slow gastric emptying
- You are unwilling or unable to self-administer a weekly subcutaneous injection
This list is not exhaustive. A complete medical history review by one of our licensed physicians is the only reliable way to determine whether retatrutide is appropriate for you.
The Regal Weight Loss Candidate Evaluation Process
When you schedule a consultation at Regal Weight Loss, our physicians review your full health history, current medications, prior weight loss attempts, metabolic lab values, and personal goals before making any treatment recommendation. If retatrutide is determined to be a strong fit, we will walk you through exactly what to expect — including dosing, potential side effects, monitoring protocols, and what results are realistic for your specific starting point. If a different medication or combination approach is a better fit, we will tell you that too. Our goal is always the right treatment for you — not simply the newest one.
Think you might be a candidate for retatrutide?
Our physicians can evaluate your eligibility and build a personalized treatment plan in a single consultation.
What to Expect: Retatrutide Treatment at Regal Weight Loss
Starting a new weight loss medication is a significant decision, and we believe every patient deserves to know exactly what their experience will look like before they begin. At Regal Weight Loss, we have built a structured, physician-supervised treatment process designed to maximize your results, minimize uncertainty, and support you at every stage of your retatrutide journey — from your very first consultation through your long-term maintenance plan.
Step 1 — Your Initial Consultation
Your retatrutide journey begins with a comprehensive one-on-one consultation with a licensed Regal Weight Loss physician. During this appointment, your provider will review your full medical history, current medications, prior weight loss history, metabolic health markers, and personal goals. This is not a quick intake form — it is a genuine clinical evaluation designed to determine whether retatrutide is the right fit for you, what starting dose makes sense, and what realistic outcomes look like for your individual profile. If retatrutide is recommended, your physician will walk you through the full treatment protocol before you leave so you know exactly what to expect.
Step 2 — Lab Work and Baseline Metrics
Before beginning retatrutide therapy, we establish a clear baseline of your metabolic health. This typically includes a comprehensive metabolic panel, fasting glucose and insulin levels, HbA1c, lipid panel, thyroid function, and body composition measurements. These baseline values serve two purposes: they help your physician personalize your treatment plan, and they give us a precise benchmark to measure your progress against as your therapy progresses. Tracking real numbers — not just the scale — is how we demonstrate the full scope of what retatrutide is doing for your health.
Step 3 — Your Personalized Retatrutide Protocol
Once your evaluation and labs are complete, your physician will build a personalized retatrutide dosing protocol tailored to your starting point and goals. Retatrutide is administered as a once-weekly subcutaneous injection, typically in the abdomen, thigh, or upper arm. Most protocols begin at a lower dose to allow your body to adjust and minimize the likelihood of gastrointestinal side effects, with gradual dose escalation over several weeks toward your target therapeutic dose. Your physician will provide complete injection training and written instructions so you feel confident administering your medication at home.
Step 4 — Ongoing Monitoring and Check-Ins
Physician supervision does not end after your first appointment at Regal Weight Loss — it is built into every phase of your treatment. You will have scheduled follow-up appointments to review your progress, assess tolerability, adjust your dose if needed, and repeat key lab markers at clinically appropriate intervals. Between appointments, our care team is available to answer questions, address side effects, and provide the support that makes the difference between patients who achieve their goals and those who do not. We monitor not just your weight but your full metabolic profile — because lasting health transformation goes beyond the number on the scale.
Step 5 — Nutrition and Lifestyle Integration
Retatrutide is a powerful tool, but it works best as part of a comprehensive approach to health. Our physicians and care team provide practical nutritional guidance designed to complement your medication — not overwhelm you with an unsustainable diet. We focus on sustainable eating habits that support lean muscle preservation, metabolic health, and long-term weight maintenance. Patients who pair retatrutide with even modest improvements in nutrition and activity consistently achieve better outcomes than those who rely on medication alone, and our team is here to help you build those habits in a way that fits your life.
Step 6 — Long-Term Maintenance Planning
Achieving your goal weight is a milestone — keeping it is the mission. Before you reach your target, your Regal Weight Loss physician will begin building your long-term maintenance strategy. This may include a planned dose reduction, a transition to a different medication, or a structured tapering protocol depending on your individual response and goals. We do not simply discharge patients when they hit a number. We build a plan that accounts for the long arc of your health — because sustainable weight loss is not a destination, it is an ongoing practice.
From your first consultation to your long-term maintenance plan, Regal Weight Loss provides the medical expertise, personalized attention, and continuous support that retatrutide therapy deserves. If you are ready to take the most advanced step available in evidence-based weight loss medicine, we are ready to guide you through it.
Ready to start your retatrutide journey?
Schedule your initial consultation today and take the first step toward the most advanced weight loss treatment available.
Retatrutide Dosing and Administration: What You Need to Know
One of the most common questions patients have before starting any injectable weight loss medication is: how does the dosing actually work? Retatrutide follows a structured dose escalation protocol — meaning you do not start at your full therapeutic dose on day one. Instead, your physician will guide you through a carefully planned ramp-up schedule designed to allow your body to adjust gradually, maximize tolerability, and ultimately reach the dose that delivers the best results for your individual profile.
How Is Retatrutide Administered?
Retatrutide is administered as a once-weekly subcutaneous injection — meaning it is injected just beneath the skin, not into a muscle or vein. The most common injection sites are the abdomen, the front of the thigh, and the upper arm. The injection is performed using a small, fine-gauge needle, and most patients report minimal to no discomfort once they are comfortable with the technique. Your Regal Weight Loss care team will walk you through proper injection technique at your first treatment appointment and provide written reference materials to take home. Injections are self-administered at home on the same day each week, making retatrutide highly convenient relative to daily medications.
Retatrutide Dose Escalation Schedule
The Phase 2 clinical trial tested retatrutide at doses of 1mg, 4mg, 8mg, and 12mg per week. In clinical practice, physicians typically begin patients at a low starting dose and escalate gradually over a period of weeks to months. A representative dose escalation schedule based on Phase 2 trial protocols looks like this:
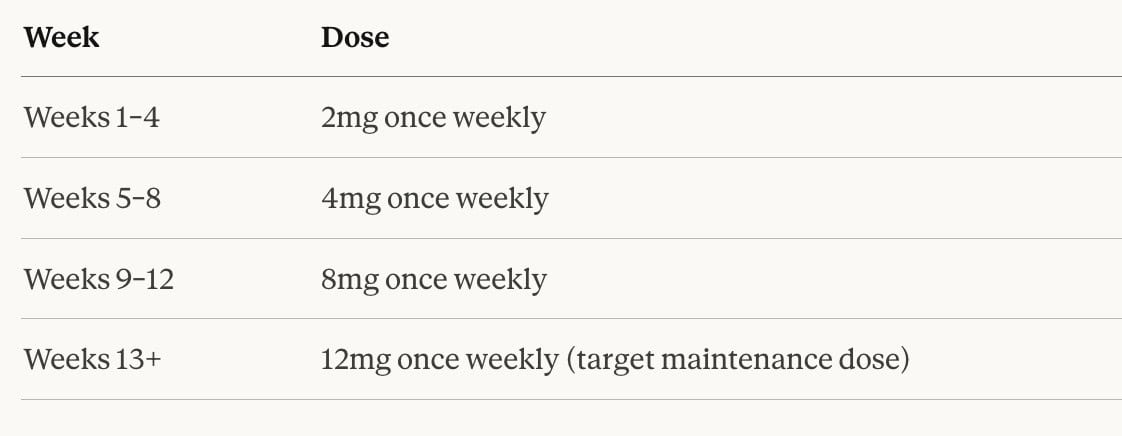
It is important to note that this schedule is a general framework — not a fixed prescription. Your Regal Weight Loss physician will personalize your escalation timeline based on how your body responds at each dose level, your tolerance for side effects, and your overall treatment goals. Some patients do exceptionally well at lower maintenance doses, and there is no clinical benefit to escalating faster than your body comfortably allows.
What Dose Produces the Best Results?
In the Phase 2 TRIUMPH trial, the highest dose group — 12mg per week — produced the greatest average weight loss at approximately 24.2% of body weight over 48 weeks. However, even lower doses produced clinically meaningful results: patients in the 4mg group lost an average of approximately 17.5% of body weight, and patients in the 8mg group lost approximately 22.8%. This dose-response relationship means that patients who cannot tolerate the highest dose still have a strong evidence base supporting meaningful weight loss at lower therapeutic doses. The goal is always the highest dose you tolerate well — not the highest dose possible.
How Long Does It Take to See Results on Retatrutide?
Most patients begin to notice reduced appetite and early weight changes within the first two to four weeks of starting retatrutide, even at the initial lower doses. Meaningful weight loss — typically defined as 5% or more of body weight — is generally observed within the first eight to twelve weeks as dosing escalates toward the therapeutic range. The most significant and sustained weight loss occurs between weeks 12 and 48 as patients reach and maintain their target dose. Unlike some medications where results plateau early, Phase 2 data showed that the weight loss curve for retatrutide had not fully plateaued by week 48 — suggesting that longer-term treatment may yield continued results beyond what the Phase 2 trial captured.
Can the Dose Be Adjusted After Starting?
Yes — and this is one of the most important advantages of working with a physician-supervised program rather than attempting to self-manage retatrutide therapy. If you experience side effects at a particular dose, your Regal Weight Loss physician can slow your escalation schedule, hold you at your current dose for a longer period, or temporarily reduce your dose to improve tolerability. Conversely, if you are tolerating your current dose well but progress has slowed, your physician may recommend escalating sooner. This kind of individualized, responsive dose management is what separates medically supervised retatrutide therapy from unmonitored access — and it is a significant factor in both safety and outcomes.
Do I Need to Refrigerate Retatrutide?
Yes. Like most injectable peptide medications, retatrutide should be stored in the refrigerator at 36°F to 46°F (2°C to 8°C) and protected from light. It should not be frozen. Unopened vials or pens should remain refrigerated until use. If you need to travel with your medication, a medical-grade insulated travel case is recommended to maintain appropriate temperature. Your Regal Weight Loss care team will provide complete storage and handling instructions when your medication is dispensed.
Retatrutide’s once-weekly injection schedule, structured dose escalation protocol, and flexible dosing range make it one of the most manageable injectable weight loss medications available — particularly under the guidance of an experienced physician. At Regal Weight Loss, we ensure every patient understands their protocol completely before they begin, and we are available throughout the process to answer questions and make adjustments as needed.
Retatrutide Side Effects: What Patients Should Know
Like all medications, retatrutide can cause side effects — and understanding them before you begin treatment is an important part of making an informed decision. The good news is that the majority of side effects observed in clinical trials were mild to moderate, most commonly occurred during the dose escalation phase, and resolved or diminished as patients adjusted to their target dose. At Regal Weight Loss, our physicians discuss potential side effects with every patient before treatment begins and monitor for them throughout the course of your therapy.
Most Common Side Effects of Retatrutide
The most frequently reported side effects in the Phase 2 TRIUMPH trial were gastrointestinal in nature — consistent with the GLP-1 class of medications. These included:
Nausea — the most commonly reported side effect, particularly during dose escalation; typically mild to moderate and transient
Vomiting — reported less frequently than nausea; usually short-lived and manageable with dietary adjustments
Diarrhea — reported in a subset of patients, most commonly during early weeks of treatment
Constipation — paradoxically, some patients experience constipation rather than diarrhea, related to retatrutide’s effect on slowing gastric motility
Decreased appetite — while this is the intended therapeutic effect, some patients experience appetite suppression more dramatically than expected, particularly at higher doses
Nausea-related fatigue — some patients report low energy during early dose escalation, typically resolving as the body adjusts
Injection site reactions — mild redness, bruising, or tenderness at the injection site, usually resolving within a day or two
How Common Are Side Effects — and Do They Go Away?
In the Phase 2 trial, gastrointestinal side effects were most prevalent during the dose escalation phase and decreased significantly once patients reached and stabilized at their maintenance dose. The structured escalation protocol used in clinical trials — and in Regal Weight Loss treatment plans — is specifically designed to minimize the severity and duration of these early side effects. Most patients who experience nausea or digestive discomfort report that it becomes manageable within two to four weeks and resolves substantially by the time they reach their target dose. Eating smaller meals, avoiding high-fat or highly processed foods, staying well hydrated, and timing injections strategically can all meaningfully reduce the likelihood and severity of gastrointestinal side effects.
Less Common but Serious Side Effects
While serious side effects were uncommon in retatrutide clinical trials, patients and physicians should be aware of the following:
- Pancreatitis — inflammation of the pancreas has been reported with GLP-1 class medications. Patients should contact their physician immediately if they experience severe, persistent abdominal pain, particularly pain that radiates to the back
- Gallbladder disease — rapid weight loss of any cause can increase the risk of gallstones; GLP-1 medications have been associated with an elevated incidence of gallbladder-related events in some studies
- Heart rate elevation — retatrutide’s glucagon receptor activation may increase resting heart rate in some patients; your physician will monitor for this during follow-up appointments
- Hypoglycemia — low blood sugar is not a primary concern for non-diabetic patients, but patients on diabetes medications may need dose adjustments as their blood sugar improves on retatrutide
- Kidney function changes — dehydration secondary to nausea, vomiting, or reduced fluid intake can affect kidney function; staying adequately hydrated is important throughout treatment
Contraindications — Who Should Not Take Retatrutide
Retatrutide is contraindicated — meaning it should not be used — in patients with the following conditions:
- Personal or family history of medullary thyroid carcinoma (MTC) — GLP-1 receptor agonists have been shown to cause thyroid C-cell tumors in rodent studies; while this has not been confirmed in humans, patients with a personal or family history of MTC should not use retatrutide
- Multiple Endocrine Neoplasia syndrome type 2 (MEN 2) — this hereditary condition is associated with elevated MTC risk and represents an absolute contraindication
- Active or history of pancreatitis — patients with a history of pancreatitis should discuss this with their physician before considering any GLP-1 class medication
- Pregnancy or planned pregnancy — retatrutide has not been studied in pregnant patients and should not be used during pregnancy; patients should discontinue retatrutide at least two months before attempting to conceive
- Severe gastrointestinal disease — patients with conditions such as gastroparesis or severe inflammatory bowel disease may not be appropriate candidates due to retatrutide’s effect on gastric motility
How Regal Weight Loss Monitors for Side Effects
Physician supervision is not a formality at Regal Weight Loss — it is the mechanism through which side effects are caught early, managed effectively, and prevented from derailing your progress. At every follow-up appointment, your physician reviews your symptom history, adjusts your dosing protocol if needed, and repeats relevant lab markers to ensure your metabolic health is moving in the right direction. Patients are also encouraged to reach out between appointments if they experience any new or concerning symptoms. Early communication with your care team is always the right move — and it is one of the most meaningful differences between a medically supervised program and unmonitored access to medication.
No medication is without risk, and retatrutide is no exception. But for the right patient, under proper physician supervision, the clinical evidence strongly supports that retatrutide’s benefits significantly outweigh its risks. The key is working with a qualified medical team that knows how to personalize your protocol, anticipate potential issues, and respond to your individual experience — which is exactly what Regal Weight Loss is built to do.
Have questions about retatrutide side effects or whether they apply to your health history?
Our physicians are available to address your specific concerns in a confidential consultation.
Frequently Asked Questions About Retatrutide
Is retatrutide FDA approved?
Retatrutide is not yet FDA approved as a standalone medication. It is currently in Phase 3 clinical trials — the final stage of the regulatory process before a formal FDA approval application can be submitted. However, retatrutide may be available to patients through licensed compounding pharmacies under physician supervision while Phase 3 trials are ongoing. At Regal Weight Loss, our physicians can evaluate whether compounded retatrutide is an appropriate option for your individual health profile.
How much weight can you lose on retatrutide?<!–?h3 >
In the Phase 2 TRIUMPH clinical trial published in The New England Journal of Medicine, participants taking the highest dose of retatrutide — 12mg per week — lost an average of 24.2% of their body weight over 48 weeks. Participants at lower doses also achieved meaningful results: approximately 17.5% at 4mg and 22.8% at 8mg. Individual results will vary based on starting weight, dose, adherence, metabolic health, and lifestyle factors. These are averages from a controlled clinical setting — your physician can help you set realistic, personalized expectations based on your specific profile.
How is retatrutide different from semaglutide (Ozempic/Wegovy)?
Retatrutide and semaglutide both activate the GLP-1 receptor, but retatrutide goes significantly further by also activating the GIP and glucagon receptors — making it a triple-receptor agonist compared to semaglutide’s single-receptor mechanism. This additional receptor activity produces greater appetite suppression, a higher resting metabolic rate, and accelerated fat breakdown that semaglutide cannot achieve. In clinical trials, retatrutide produced approximately 24.2% average body weight loss compared to semaglutide’s approximately 15% — in a shorter trial duration.
How is retatrutide different from tirzepatide (Mounjaro/Zepbound)?
Both retatrutide and tirzepatide activate the GLP-1 and GIP receptors. The critical difference is that retatrutide additionally activates the glucagon receptor — a third pathway that increases the body’s resting metabolic rate and accelerates the breakdown of stored fat in ways tirzepatide cannot. This third receptor is believed to be responsible for the additional weight loss advantage retatrutide demonstrated in clinical trials — approximately 24.2% average body weight loss versus tirzepatide’s approximately 20% — achieved in a shorter timeframe.
Can I get retatrutide if it is not FDA approved yet?
Yes, in some cases. Retatrutide may be available through licensed compounding pharmacies, which are regulated by state pharmacy boards and the FDA and are legally permitted to compound medications for patient use under physician supervision. This is the same pathway through which compounded semaglutide and tirzepatide have been available to patients. Access through a compounding pharmacy requires a valid prescription from a licensed physician — which is why working with a qualified medical provider like Regal Weight Loss is essential. Our physicians will evaluate your eligibility and, if appropriate, provide the necessary prescription and ongoing supervision.
What are the most common side effects of retatrutide?
The most commonly reported side effects of retatrutide in clinical trials were gastrointestinal in nature, including nausea, vomiting, diarrhea, and constipation. These side effects were most prevalent during the dose escalation phase and diminished significantly as patients stabilized at their maintenance dose. The structured dose escalation protocol used at Regal Weight Loss is specifically designed to minimize the severity and duration of these early side effects. Serious side effects were uncommon in clinical trials but include pancreatitis, gallbladder events, and elevated heart rate, which your physician will monitor for throughout your treatment.
How long does it take for retatrutide to work?
Most patients begin to notice reduced appetite within the first two to four weeks of starting retatrutide, even at the initial lower starting doses. Measurable weight loss — typically 5% or more of body weight — is generally observed within the first eight to twelve weeks as the dose escalates toward the therapeutic range. The most significant and sustained weight loss typically occurs between weeks 12 and 48. Importantly, the Phase 2 trial data showed that the weight loss curve had not fully plateaued by week 48, suggesting that longer-term treatment may produce continued results beyond what the trial captured.
How often do you inject retatrutide?
Retatrutide is administered as a once-weekly subcutaneous injection — meaning once per week, on the same day each week, injected just beneath the skin. Common injection sites include the abdomen, front of the thigh, and upper arm. The once-weekly schedule makes retatrutide highly convenient compared to daily medications and is consistent with the administration schedules of semaglutide and tirzepatide. Your Regal Weight Loss care team will provide complete injection training and written instructions before you begin self-administering at home.
Is retatrutide safe?
Based on Phase 2 clinical trial data, retatrutide demonstrated an acceptable safety profile across all dose groups, with the majority of side effects being mild to moderate and gastrointestinal in nature. No unexpected safety signals were identified in Phase 2 that have prevented progression to Phase 3 trials. That said, as with any medication — particularly one still in clinical development — long-term safety data continues to be gathered through ongoing Phase 3 trials. Working with a physician-supervised program like Regal Weight Loss ensures that your individual safety profile is evaluated before treatment begins and monitored continuously throughout your therapy.
Can I take retatrutide if I have type 2 diabetes?
Retatrutide may be appropriate for patients with type 2 diabetes, and in fact the Phase 2 trial included participants with weight-related comorbidities including diabetes. Because retatrutide improves insulin sensitivity and blood sugar regulation, patients with type 2 diabetes who are also taking diabetes medications — particularly insulin or sulfonylureas — may need dose adjustments to those medications as their blood sugar improves. This is one of the most important reasons physician supervision is essential: your Regal Weight Loss provider will coordinate your retatrutide therapy with your overall diabetes management plan to ensure both safety and optimal outcomes.
Will I gain weight back if I stop taking retatrutide?
Weight regain after discontinuing GLP-1 class medications is a well-documented phenomenon — and retatrutide is expected to follow a similar pattern based on what is understood about the mechanism of action. These medications work by modulating hunger hormones and metabolic pathways that, without ongoing support, tend to return toward baseline. However, the degree of regain varies significantly between patients and is strongly influenced by the lifestyle habits, nutrition patterns, and metabolic improvements developed during treatment. At Regal Weight Loss, we begin planning your long-term maintenance strategy before you reach your goal weight — including whether a reduced maintenance dose, a transition to a different medication, or a structured tapering plan is the right approach for you.
How much does retatrutide cost?
The cost of retatrutide through a compounding pharmacy varies depending on the dose, the pharmacy, and your geographic location. Because retatrutide is not yet FDA approved, it is not currently covered by most insurance plans. At Regal Weight Loss, we are transparent about all costs associated with your treatment — including medication, physician visits, and lab work — before you begin. We encourage you to schedule a consultation to receive a complete picture of what your personalized retatrutide program would involve and what the associated investment looks like for your specific dose and protocol.
Is retatrutide available near me?
Retatrutide is available through Regal Weight Loss to patients who qualify following a physician evaluation. Because our program includes telehealth consultation options, geographic proximity is not a barrier for most patients. Contact our team to confirm availability in your area and to schedule your initial eligibility consultation.
Take the First Step Toward the Most Advanced Weight Loss Treatment Available
You have done the research. You understand what retatrutide is, how it works, what the clinical evidence shows, and what a physician-supervised treatment program looks like. Now the only remaining question is whether retatrutide is the right next step for you specifically — and that is exactly what a Regal Weight Loss consultation is designed to answer.
Why Patients Choose Regal Weight Loss for Retatrutide
There is no shortage of places offering access to weight loss medications. What is far less common is a program that combines genuine physician oversight, personalized dosing protocols, comprehensive metabolic monitoring, and long-term maintenance planning into a single, cohesive patient experience. At Regal Weight Loss, retatrutide is not simply dispensed — it is prescribed thoughtfully, monitored continuously, and supported by a care team that measures success not just in pounds lost but in lasting health transformation. That distinction matters — and for patients who have tried other approaches without the results they deserve, it makes all the difference.
✓ Licensed physicians with expertise in obesity medicine
✓ Comprehensive evaluation before any treatment is recommended
✓ Personalized dosing protocols tailored to your health profile
✓ Ongoing monitoring and physician access throughout treatment
✓ Transparent pricing — no hidden fees, no surprises
✓ Telehealth options available for patient convenience
What Happens When You Schedule a Consultation?
Booking a consultation at Regal Weight Loss is the beginning of a process — not a commitment to any particular treatment. During your initial appointment, one of our medical assistants will review your health history, discuss your goals, and give you an honest, evidence-based assessment of whether retatrutide or another treatment approach is the right fit for you. You will leave with a clear picture of your options, your personalized protocol if retatrutide is recommended, and a full understanding of what your treatment investment looks like. There is no pressure, no obligation, and no guesswork — just a straightforward medical conversation with a physician who is genuinely invested in your outcome.
Ready to Find Out If Retatrutide Is Right for You?
Schedule your retatrutide consultation today. Spots are limited — our centers take on a limited number of new retatrutide patients each month to ensure every patient receives the attention and oversight their treatment deserves.
